Heart Transplant Program

Leaders in Heart Transplantation
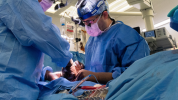
The Stanford Heart Transplant Program is one of the most active and successful heart transplant centers in the country. Our transplant program exceeds the national average for survival post heart transplantation. We pioneer innovations in heart transplantation so we can provide more hearts to more people, often with shorter-than-expected wait times.
300 Pasteur Drive
2nd Floor, Room A21
Stanford,
CA
94305
Phone: 650-723-5468
What We Offer You for Heart Transplantation
- One of the largest programs in the U.S. and the largest in the Bay Area, with specialized expertise in heart transplants and comprehensive care for advanced heart failure and its underlying causes. Go to Conditions Treated
- Advanced treatment options, including multiorgan transplants, mechanical heart devices, and lifelong follow-up care. Go to Treatments
- Outstanding quality and outcomes in heart transplantation, with survival rates better than the national average at one year after heart transplant. Go to Quality and Outcomes
- Team approach, with highly experienced specialists who create a comprehensive care plan customized to your needs. Go to Your Care Team
- Clinical trials that provide eligible patients access to medical therapies, devices, and diagnostic testing to improve outcomes, prevent complications, and improve quality of life after transplant. Go to Clinical Trials
- Comprehensive support services to help you, your family, and your support network focus on health and healing.
Go to Support Services - Easily accessible care, with transplant coordinators who guide you through the process and help with travel, lodging, and other needs during your stay.
Go to Accessible Care
Heart disease and other conditions can weaken the heart, leading to heart failure. Treatments, such as medications, medical devices, and surgery, can often manage these conditions and delay or prevent heart failure.
But even with treatment, some people may experience worsening heart failure which severely limits their daily activities. When all available therapies with medicines and devices have failed, we may recommend an evaluation for advanced therapies such as heart transplantation.
Our heart transplant team includes heart surgeons, cardiologists, and other specialists to provide a comprehensive assessment of your health. Working together, we recommend the best treatment options for you, which may include a heart transplant.
Conditions that can lead to severe heart failure and ultimately require a heart transplant include:
Stanford Health Care achieves exceptional heart transplant outcomes, with one-year survival rates of 95%—better than national averages.
Stanford Health Care has earned worldwide recognition as a pioneering center for heart transplants. Since 1968, when our team performed the first successful adult human heart transplant in the U.S., and in 1981, when our team performed the first sucessful heart-lung transplant, our specialists have advanced the field. Today, we pursue groundbreaking research to improve care for people receiving heart transplants.
Innovation Highlights
Stanford Health Care accomplishments in heart transplantation include:
- A noninvasive blood test to replace a heart biopsy to check for organ rejection, a condition in which the immune system attacks the transplanted heart
- New immunosuppressant medications that reduce the body’s immune response to help prevent rejection of the transplanted heart
- New techniques to obtain and transport donor hearts, providing more hearts for more people and reducing wait times while maintaining excellent outcomes
Photography by Winston L. Trope
Advanced Diagnostics
Our team provides a thorough evaluation to make sure a heart transplant is the right option for you.
Diagnostic tests also help us monitor your health as you wait for a donor heart. How long you are on the transplant waitlist can vary, depending on many factors. Your doctor will explain more about the waitlist.
Based on your specific health needs, you may need one or more tests, such as:
- Blood tests to check for signs of diabetes, high cholesterol, and other conditions
- Blood and tissue typing to help with donor matching and preventing organ rejection
- Imaging, such as a chest X-ray, ultrasounds, and CT scans
- Heart tests, such as:
- Cardiac catheterization
- Echocardiogram (echo)
- Electrocardiogram (EKG or ECG)
- Stress test
- Psychosocial evaluation to identify and address issues that affect transplantation and recovery, such as family support, financial resources, and stress
Shorter Wait Times
The wait time for undergoing a heart transplant at Stanford Health Care is better than other heart transplant centers in the U.S. Some of the sickest patients received transplants in as little as a week. The shorter wait times are related in part to a new system initiated by the United Network for Organ Sharing (UNOS).
Shorter wait times help improve the chances of successful heart transplantation, because people receive a new heart while they are stronger and healthier.
We transplanted more patients, we transplanted them faster, and we have been getting great outcomes.
Treatments
Your care team includes specialists with years of experience treating heart failure and underlying conditions. Our heart transplant team has expertise in:
- Heart transplant
- Multiorgan transplants including heart/lung, heart/kidney, and heart/liver
- Left ventricular assist device (LVAD) implantation, as a bridge-to-transplant or destination (long-term) therapy
Clinical Trials
Heart Transplant
0
Open Clinical Trials
View All »
Clinical trials are research studies that evaluate a new medical approach, device, drug, or other treatment. As a Stanford Health Care patient, you may be eligible to participate in clinical trials to evaluate these new treatments.
Open trials refer to studies currently recruiting participants or that may recruit participants in the near future. Closed trials are not currently enrolling, but similar studies may open in the future.
Excellence in Quality and Outcomes
Stanford Health Care’s Heart Transplant Program is one of the most active and successful transplant centers in the country, allowing more patients to receive a heart transplant with positive outcomes. You will benefit from:
- Fastest heart transplantation: Stanford Health Care’s program allows for a heart transplant sooner, sometimes within weeks of being listed. The adult heart transplant program has the shortest wait time in California and is top 5 in the nation.1
- Higher organ availability: Stanford Health Care’s organ acceptance ratio is top 10 in the nation.1
- Excellent survival rates: Heart transplantation survival rates are better than the national average after the heart transplant.
|
1
|
1
Stanford Health Care Rates |
1
National Average Rates |
|---|---|---|
|
1
Patient 1-year survival post-transplant |
1
93.00%1 |
1
91.57%1 |
|
1
Graft (heart) 1-year survival post-transplant |
1
92.27%1 |
1
91.24%1 |
These results speak to the consistency and thoroughness with which we care for patients over time. To learn more about statistics related to heart transplantation, visit the Scientific Registry of Transplant Recipients.
Our Patients
Read a patient story: Another chance at life, heart-liver transplantations
Read a patient story: Double transplant at Stanford saves life of critically ill COVID-19 patient
Your Heart Transplantation Team
Our heart transplant team includes specialists from multiple disciplines for outstanding care, education, and support throughout your transplant process. You will receive support from a team of specially trained nurses, care coordinators, and other providers. We work closely with your referring physician during your evaluation and treatment at Stanford Health Care.

Your Providers
Cardiovascular Transplant Surgeons
Transplant surgeons are doctors who specialize in organ transplantation. They evaluate you before putting you on the transplant list, perform the transplant surgery, and oversee your care in the hospital after transplant.
Our cardiovascular transplant surgeons specialize in using surgery and minimally invasive procedures to treat heart and vascular (blood vessel) conditions. These doctors perform surgeries on the heart and blood vessels. They also implant ventricular assist devices (VADs) to support the heart.
View All {0} Cardiothoracic Surgeons »Heart Failure Cardiologists
Heart failure cardiologists have advanced training in diagnosing and treating heart failure and managing people who have VADs. After a physical exam and specialized tests, such as an echocardiogram, they determine your treatment plan and oversee your care.
View All {0} Heart Failure Cardiologists »Immunocompromised Infectious Disease Specialists
Infectious disease specialists are doctors who diagnose, treat, and help prevent diseases caused by bacteria, viruses, fungi, and parasites in transplant recipients. People who receive organ transplants must take medications that suppress the immune system (immunosuppressants) to help prevent organ rejection. Immunosuppressants increase your risk of developing infections, and an infectious disease specialist helps minimize that risk.
View All {0} Infectious Disease Doctors »Cardiac Anesthesiologists
Cardiac anesthesiologists are doctors with training to administer and manage anesthesia during a surgical procedure, such as organ transplantation. They are also responsible for managing and treating changes in your critical life functions—breathing, heart rate, and blood pressure—during surgery.
View All {0} Anesthesiologists »Advanced Practice Providers
Advanced practice providers include nurse practitioners and physician assistants. They work with transplant surgeons and heart failure cardiologists to manage your care before and after the transplant.
View All 3 Advanced Practice Providers »Stanford is an Academic Medical Center, which is a type of hospital setting where doctors teach the entire spectrum of medical education. Students range from beginning medical students to fully licensed and practicing doctors completing advanced sub-specialty training. Stanford Medicine is a partnership between Stanford University School of Medicine and Stanford Health Care. Since Stanford is a teaching hospital, you can expect to meet many providers and providers in training.
- Attending Physician: a doctor who supervises doctors in training or in medical school
- Fellow: a doctor doing postgraduate level work and specializing in care of patients with specific conditions
- Resident: a doctor who has graduated from medical school and is in training (also called “residency”) here at Stanford. A resident is also called an intern
- Medical Student: a student who is currently enrolled in medical school with the goal of becoming a doctor

Extended Care Team
Anna DePena Haye, Physician Assistant, APP Manager
Heather Packard, RN, Heart Transplant and LVAD Program Manager
Pre-transplant nurse coordinators manage your evaluation process once you are referred to us for organ transplantation. They help you understand the physical and psychosocial implications of a transplant. They also obtain your medical history and testing from your doctors, and inform you about upcoming appointments and tests. They monitor your health while you are on the organ waitlist before transplant surgery.
Nicole Cote, RN
Clara Resurreccion, RN
Pre-transplant patient care coordinators are your first point of contact. They schedule your appointments with the team for evaluation, tests, and other services to complete the evaluation process. Pre-transplant patient care coordinators work closely with pre-transplant nurse coordinators to help you navigate the entire evaluation process.
Val Cannon
Post-transplant nurse coordinators organize all aspects of your care after your transplant. Before you leave the hospital, they provide you with the education you need to have a successful post-transplant course. They manage your outpatient care when you come to the clinic and are your primary contact for your post-transplant needs.
Karin Choy, RN
Lori Fox, RN
Stefannie Panlilio, RN
Rebecca Wainwright, RN
Jennifer Piazza, RN
Along with your post-transplant nurse coordinator, post-transplant patient care coordinators help coordinate your post-transplant care. They schedule your clinic appointments and tests, track test results, and manage your medication refills.
Joyce Rowan, MA
Yasmin Fonseca-Lopes, MA
Hannah Miller, RN, BSN, MSN
Roma Patel, PA-C, RN
Omar Odeh, RN
Jesstine Carmona, MA
Registered dietitians work closely with you and your family to help you meet your nutritional needs before and after the transplant. Transplant dietitians help you learn about a nutritious diet to speed your recovery and maintain lifelong health.
Physical therapists help you regain your strength and function with movement and endurance exercises after the transplantation.

Support Services
We offer a vast array of support services to help you feel your best throughout your care, from diagnosis through treatment and recovery.
Our heart transplant team provides you with comprehensive services at the Stanford Health Care main campus. You can access all your care—from diagnostic evaluation to treatment to follow-up care—in one place.
For Referring Physicians
PHYSICIAN HELPLINE
Fax: 650-320-9443
Monday–Friday, 8 a.m.–5 p.m.
Stanford Health Care provides comprehensive services to refer and track patients, as well as the latest information and news for physicians and office staff. For help with all referral needs and questions, visit Referral Information.
You may also submit a web referral or complete a referral form and fax it to 650-320-9443 or email the Referral Center at ReferralCenter@stanfordhealthcare.org.
International Patients
Phone: +1 650-723-8561
Email: IMS@stanfordhealthcare.org
1 “Heart, Stanford Health Care,” Scientific Registry of Transplant Recipients, accessed January 12, 2024